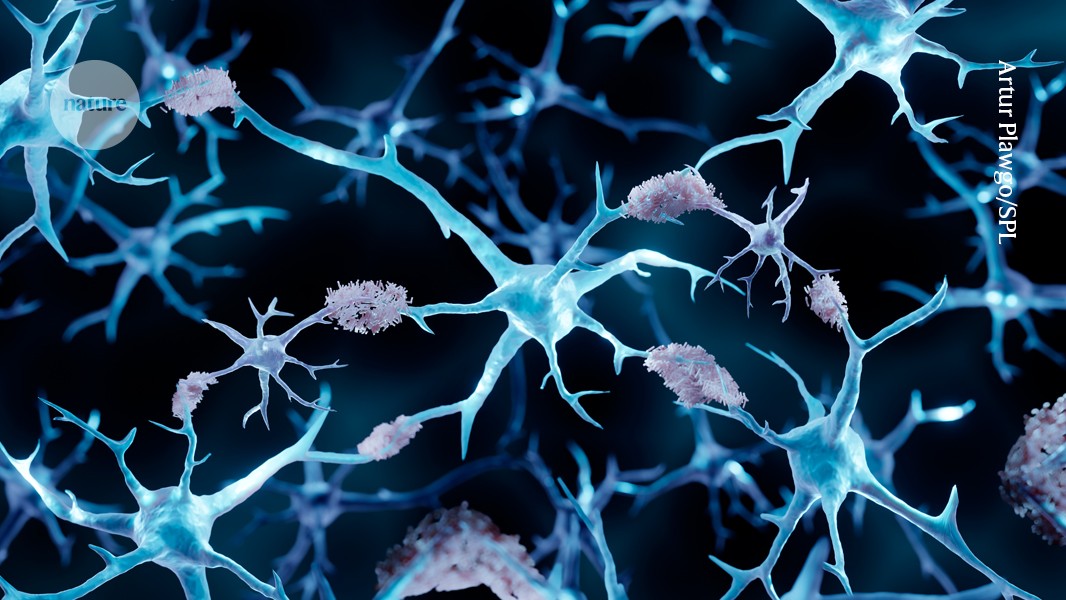
For decades, epidemiological data has suggested an inverse correlation between the incidence of cancer and Alzheimer's disease in the same patients, fueling speculation regarding underlying biological interactions. A recent study published in the journal Cell provides molecular evidence supporting this long-observed phenomenon.
This research focuses on identifying the specific mechanism driving this protective effect, potentially involving cellular stress responses or metabolic pathways common to both conditions. The findings suggest that processes activated in one disease state might inadvertently suppress the pathology of the other.
Specifically, the investigation centered on a protein pathway implicated in cellular survival and proliferation, which appears to be upregulated in certain cancer contexts. This upregulation, according to the report, may confer resistance against the amyloid-beta plaque accumulation characteristic of Alzheimer's pathology.
Data cited in the Nature analysis points to prior work, including research by Ospina-Romero et al. in JAMA Network Open, which documented population-level observations regarding these competing pathologies. The new molecular findings offer a mechanistic explanation for these broad statistical trends.
Understanding this shared molecular architecture could significantly influence drug development strategies for neurodegenerative disorders. If a pathway protects against Alzheimer's, modulating it could offer therapeutic benefits without necessarily inducing oncogenic risk, provided the balance is correctly managed.
However, the complexity of both diseases mandates caution, as cancer cells often exhibit high metabolic activity that differs significantly from the quiescent state often seen in aging neurons. Further validation is required to assess the translational viability of targeting this specific protein in human clinical settings.
The broader implication for global health economics is substantial, given the escalating costs associated with managing Alzheimer's disease worldwide. Identifying a robust protective factor could reshape long-term care investment strategies.